Dr. Eshika Dubey
The human microbiota consists of a diverse range of microorganisms—bacteria, fungi, viruses, protozoa, archaea, and yeast—predominantly made up of bacteria. These microbes inhabit various sites in the body, such as the gut, oral cavity, respiratory tract, and skin. Upon birth, commensal bacteria begin colonizing the host, gradually developing into a complex and dynamic ecosystem as the host matures. These host-microbe interactions evolve into mutually beneficial relationships, where symbiotic bacteria play vital roles in metabolism, immune function, nutrient provision, defense mechanisms, and the formation of the intestinal structure. They synthesize essential vitamins, support immune responses, and contribute to the integrity of the mucosal immune system. Disruptions in this microbial ecosystem, often induced by external factors, can lead to various diseases, including cardiovascular diseases (CVDs), cancer, diabetes, inflammatory bowel disease (IBD), respiratory and brain disorders, as well as chronic kidney and liver diseases.
Recent studies have highlighted the significant connection between gut microbiota and gastrointestinal (GI) cancers. The GI tract, with its conducive environment for microbial survival, serves as a critical site for microbiota-host interactions that can influence disease development. Dysbiosis—an imbalance or disturbance in the microbiota—can activate inflammatory pathways in the GI mucosa, triggering oxidative stress, the release of pro-inflammatory cytokines, nitric oxide (NO), and cyclooxygenase-2 (COX-2) production. This dysbiosis can contribute to the onset of GI cancers through mechanisms like epithelial-to-mesenchymal transition (EMT), the accumulation of NKT cells in bile metabolism, or the secretion of inflammatory mediators, as seen in hepatocellular carcinoma (HCC). Helicobacter pylori, for example, disrupts epithelial cell homeostasis and plays a pivotal role in gastric cancer by inducing immunosuppression, DNA damage, cell proliferation, neovascularization, and genomic instability—key drivers of carcinogenesis.
Leveraging microbiota biomarkers for diagnostic purposes, in conjunction with traditional screening methods, holds promise for early detection and improved cancer management. Microorganisms such as Fusobacterium nucleatum, Bacteroides fragilis, Streptococcus bovis, Citrobacter species, Enterococcus faecalis, Porphyromonas species, and Slakiahave been identified as potential biomarkers for colorectal cancer (CRC), adenomatous polyps, and other GI cancers. Microbe-derived metabolites in stool or serum also show potential as diagnostic tools, with serologic tests for Helicobacter pylori antibodies, pepsinogen levels, and nitrosating/nitrate-reducing bacteria aiding in investigation. Additionally, the gut microbiota plays a crucial role in drug metabolism, impacting both efficacy and side effects.
The microbiota also serves as a predictor of treatment responses, particularly by reducing the toxicity of conventional cancer therapies. Modulating gut microbial composition—through beneficial strains such as Lactobacillus, Bifidobacterium, Faecalibaculum rodentium, and Streptococcus thermophiles—has shown promise in improving cancer treatment outcomes. Furthermore, the gut microbiome regulates immune responses to immunotherapy. Preclinical and observational data suggest that a diet rich in fiber and fermented foods can influence immunity and inflammation, enhancing the effectiveness of targeted immunotherapies.
The microbiota exerts dual effects in the context of gastric cancer. While dysbiosis accelerates cancer development, technological interventions targeting the microbiota may offer novel approaches to diagnosis and treatment. This review seeks to explore both aspects—understanding the role of microbiota in gastric cancer progression and investigating potential microbiota-based therapeutic strategies.
Mechanisms by Which Microbiota Influence GI Cancer Development
Understanding the mechanisms by which gut microbiota influence gastrointestinal (GI) cancer development is essential for advancing treatment strategies. By adapting to the unique microenvironments within the body, bacteria contribute to numerous processes that can affect disease susceptibility. Over time, microbial species undergo genetic changes through mutations and horizontal gene transfer, influencing health outcomes and disease susceptibility.
Procarcinogenic microorganisms such as Bacteroides fragilis, Fusobacterium nucleatum, and Escherichia coli can accelerate cancer development, particularly in the colon. For example, Escherichia coli induces DNA damage, promoting the progression of colon cancer. Microbial colonization begins at birth, playing a pivotal role in immune system development and regulation throughout life. By modulating immune responses, microbiota can influence cancer progression. For instance, Fusobacterium nucleatum suppresses T-cell activity, impairing the immune response and fostering an environment conducive to cancer progression. Studies show that high levels of Fusobacterium nucleatum are inversely correlated with the presence of CD3+ T-cells, further indicating its role in immune suppression.
Dysbiosis—the imbalance in the microbiome—has been associated with various diseases, including cancer. It can alter immune responses, metabolic processes, and gut barrier integrity, all of which exacerbate disease severity. Dysbiosis creates a pro-inflammatory environment in the GI tract, supporting tumor growth and metastasis. It also impairs immune function, reducing the body’s ability to destroy cancer cells, while enhancing the metabolism of carcinogens and weakening the mucosal barrier, which facilitates DNA damage and further genetic mutations.
The production of bacterial metabolites plays a significant role in cancer initiation. In colorectal cancer, for example, the microbiota ferments complex carbohydrates, producing short-chain fatty acids (SCFAs) such as acetate, propionate, and butyrate. These SCFAs communicate with host cells and influence cellular processes. Butyrate, in particular, serves as an energy source for colonocytes, but in tumor cells, it inhibits proliferation by modulating gene expression and downregulating cell cycle regulators like p21 and p27. This explains the beneficial effects of butyrate in cancer.
Microbial toxins also contribute to carcinogenesis. Pathogenic bacteria produce protein toxins that directly damage DNA, leading to genetic instability and promoting tumor formation. These toxins may alter cell signaling pathways, encouraging uncontrolled cell proliferation and obstructing apoptosis, which creates a favorable environment for tumor growth.
Impact of Microbiota on Cancer Outcomes
The human microbiota profoundly influences the tumor microenvironment, immune responses, and treatment efficacy, making it a critical factor in cancer outcomes. In colorectal cancer (CRC), specific bacteria like Fusobacterium nucleatumcan be detected in fecal samples from high-risk individuals, including those with adenomas or early-stage CRC. These microbes, along with their metabolic and genotoxic products, offer promising biomarkers for early cancer detection.
The microbiome is also emerging as a predictive biomarker for treatment responses. It influences the metabolism of chemotherapeutic agents, their efficacy, and potential side effects. For example, the gut microbiota affects the metabolism of chemotherapy drugs like irinotecan, oxaliplatin, and 5-fluorouracil, all commonly used to treat metastatic CRC. The microbiota can also modulate chemotherapy toxicity, such as causing irinotecan-induced diarrhea by reactivating the drug’s active metabolite through bacterial β-glucuronidases. Targeting these enzymes could mitigate such side effects.
In addition, the gut microbiome impacts the effectiveness of immunotherapy. Studies on melanoma patients undergoing checkpoint inhibitor therapy have shown that responders exhibit a higher abundance of beneficial bacteria like Faecalibacterium and Ruminococcaceae. Radiation therapy and chemotherapy also interact with the microbiome, altering its composition and potentially affecting treatment efficacy. For instance, Bacteroides and Enterobacteriaceae increase following radiation, while beneficial bacteria like Faecalibacterium prausnitzii and Bifidobacterium decrease, potentially affecting therapeutic outcomes.
Strategies to Modulate Microbiota for Cancer Prevention and Treatment
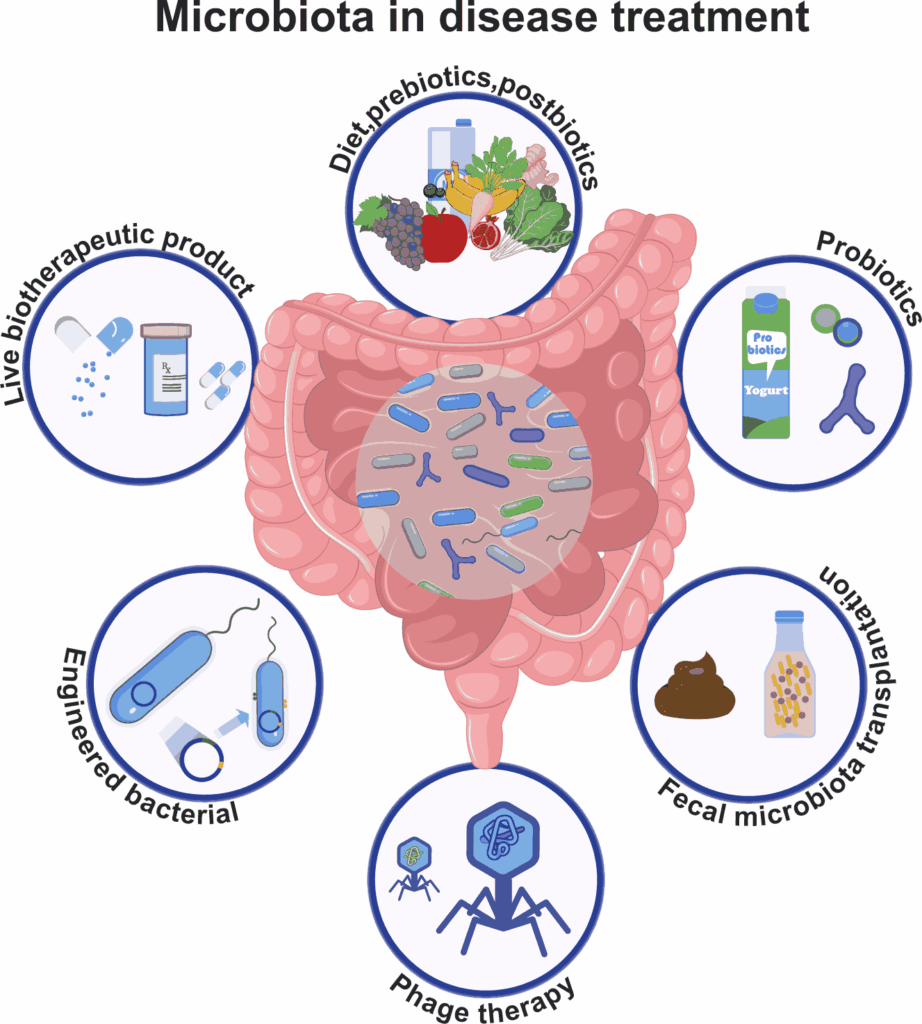
Given the significant role of the microbiota in cancer development, modulation of the microbiome offers a promising strategy for cancer prevention and treatment. Prebiotics and probiotics are potential interventions in GI cancer management. Probiotics—live microorganisms that confer health benefits—can help modulate gut microbiota and prevent cancer development. In animal models, probiotics such as Lactobacillus rhamnosus and Escherichia coli Nissle 1917 have demonstrated protective effects against colorectal cancer by reducing tumor size and preventing inflammation.
Prebiotics, which promote the growth of beneficial gut microbes, can also be beneficial. By enhancing probiotic activity, prebiotics help reduce inflammation, support gut barrier integrity, and prevent pathogenic colonization, ultimately lowering the risk of GI cancers. The SCFAs produced by probiotic fermentation—such as butyrate, acetate, and propionate—help protect the gut epithelium and suppress tumor growth.
Fecal microbiota transplantation (FMT) is another promising approach for restoring microbial balance in cancer patients. By transferring healthy gut microbiota from a donor to a patient, FMT can correct dysbiosis and improve treatment outcomes. However, careful implementation is required to minimize risks and maximize therapeutic benefits.
Bacteriophage-based strategies also offer potential for microbiome modulation in cancer treatment. Bacteriophages can target specific bacteria, alter their surface characteristics, and make them more susceptible to therapy. This targeted approach could be particularly useful in modulating the microbiome to enhance cancer treatments.
While conventional antibiotics have been linked to poor cancer outcomes due to their impact on microbial diversity, selective use of antibiotics against pathogenic bacteria may help prevent cancer in high-risk individuals or improve outcomes for cancer patients.
Effective Strategies to Modulate Microbiota in GI Cancer
The gut microbiota plays a crucial role in the effectiveness of anticancer treatments for gastrointestinal (GI) cancers. Under normal conditions, the immune system maintains a balance between responding to harmful microorganisms and tolerating non-pathogenic antigens and beneficial microbiota, which are essential for immune homeostasis. This balance is mediated primarily by T lymphocytes, particularly regulatory T cells (Tregs), which are involved in cellular immunity and the initiation of antigen-specific immune responses.
Given that the gastrointestinal system has the largest mucosal surface area in the body, it is also a major reservoir of antigens. Disruption of this microbiota balance, as seen in gastrointestinal malignancies, can lead to impaired chemotherapy responses and hinder cancer treatment effectiveness.
Several strategies have been proposed to enhance the efficacy of anticancer therapies in patients with gastrointestinal cancer by modulating the microbiota. These include:
- Nutritional Interventions: Supplementing microbiota-derived metabolites such as indole-3-acetic acid (IAA), a tryptophan metabolite, has been shown to improve chemotherapy outcomes in GI cancer patients. Higher levels of IAA in these patients correlate with better responses to anticancer drugs. IAA is thought to stimulate the release of myeloperoxidase, an enzyme from neutrophils. This enzyme oxidizes IAA, which, when combined with chemotherapy agents, reduces the activity of enzymes that degrade reactive oxygen species (ROS). This results in the accumulation of ROS, leading to the breakdown and destruction of cancer cells, thus inhibiting their growth and proliferation.
- Probiotics: The role of probiotics in modulating the host’s immune response, preventing pathogen colonization in the GI tract, and enhancing gastrointestinal barrier integrity is increasingly recognized in cancer treatment. Probiotics have also been shown to promote the apoptosis (programmed cell death) of cancer cells in GI malignancies, thus inhibiting their growth and proliferation. Studies have demonstrated significant efficacy, particularly in colorectal cancer, where probiotics decreased cancer cell viability by up to 78%.
- Anti-Angiogenic Strategies: Cancerous tumors often produce pro-angiogenic factors such as vascular endothelial growth factor A (VEGF-A), which supports tumor growth by stimulating the formation of new blood vessels. These factors also attract regulatory T cells (Tregs) to the tumor site, which suppress the immune response, enabling cancer cells to evade detection and continue proliferating. Supplementation with anti-angiogenic factors, such as antibodies targeting VEGF-A or hepatocyte growth factor (HGF), can reduce the accumulation of Tregs and thus enhance the immune system’s ability to target cancer cells. For instance, the anti-HGF antibody rilotumumab has been shown to reduce circulating Tregs in patients with gastric cancer, providing a promising avenue for improving cancer treatment.
- Fecal Microbiota Transplantation (FMT): FMT, a rapidly advancing biotherapeutic intervention, involves transferring fecal material from a healthy donor to the gastrointestinal tract of a patient with cancer. This approach aims to restore microbial balance (dysbiosis) and enhance the response to chemotherapy. Studies in animal models have demonstrated that FMT can improve the host’s resistance to cancer by modifying the microbiota to favor an environment more conducive to treatment efficacy.
- Immune Checkpoint Inhibitors: Immune checkpoint inhibitors, such as anti-CTLA4, anti-PD-1, and anti-PD-L1, have been shown to improve the efficacy of anticancer therapies, including in GI cancers. These inhibitors work by blocking immune checkpoints that cancer cells use to evade immune detection, allowing the immune system to mount a more effective response against the tumor.
While these strategies have shown promise, their clinical application is not without challenges. Adverse effects, complications, and limitations persist, necessitating further exploration of interventions to ensure safety and minimize risks. Ongoing research is required to optimize these strategies for broader clinical use.

Opportunities and Future Directions
In recent years, there has been a surge in research focused on the gut microbiome, driven by advancements in high-throughput sequencing technologies such as 16S rRNA sequencing and shotgun sequencing. As microbiome research continues to evolve, emerging technologies like metatranscriptomics, metabolomics, culturomics, and synthetic biology offer new opportunities to understand the complex interactions between microbiota and host health.
Next-Generation Technologies:
- Culturomics enables the cultivation of a wide variety of bacteria from stool samples by creating micro-chambers with unique culture conditions. This high-throughput approach significantly enhances our ability to study bacterial diversity and functionality in the human microbiome.
- Metagenomics and Shotgun Sequencing provide detailed, culture-free analyses of microbial communities by sequencing all genetic material in a sample. These techniques can identify phylogenetic markers and microbial functions that influence health and disease.
Advanced Models in Microbiome Research:
- Tools like the Simulator of the Human Intestinal Microbial Ecosystem (SHIME®), Human Microbial X (HuMix), and the Rapid Assay of Individual Microbiome (RapidAIM) offer powerful systems for studying human gut microbiota in vitro, providing insights into microbial dynamics and their impact on disease processes.
- With the ability to modify the microbiome, new interventions, both broad (e.g., prebiotics, antibiotics, FMT) and targeted (e.g., bioengineered probiotics), hold great potential for disease prevention and treatment.
The Role of Biotech and Computational Biology: The integration of computational biology with microbiome research is crucial for interpreting large-scale genomic data. Biotech companies play a vital role in advancing microbiome studies, offering tools for data generation and analysis. Collaboration between research institutions, hospitals, and industry stakeholders is essential for improving pathogen control, understanding antimicrobial resistance, and refining treatment strategies.
Collaborative Efforts in Microbiome Research:
- Public-Private Partnerships: Collaboration between government, non-profit organizations, and private industries is crucial for advancing microbiome-based therapies, including microbiome medicines and probiotics, ensuring their safety, efficacy, and accessibility.
- Global Initiatives: International guidelines like CONSORT, STROBE, and MIMARKS aim to standardize microbiome research and improve the quality of studies, providing a framework for the reliable reporting of findings.

As microbiome research progresses, it is essential to continue investing in both financial and intellectual resources to develop safe and effective microbiome-based therapies for gastrointestinal cancer treatment. The application of advanced technologies and cross-disciplinary collaborations will pave the way for new, targeted interventions.
Conclusion
The stability of the human microbiota ecosystem is essential for maintaining health. However, disruptions to this balance, known as dysbiosis, are associated with a range of diseases, including cancer. Studies have shown that alterations in the gut microbiota can influence cancer development and response to treatment, highlighting the importance of microbiome-based strategies in cancer care.
The gut microbiota influences cancer therapy by acting as a facilitator of treatment responses, enhancing detoxification from anticancer drugs, modulating immune responses, and improving the outcomes of immunotherapies and radiation treatments. Disruption of the microbiota can lead to cancer-promoting changes in the host, including genetic damage, immune suppression, and the production of carcinogenic metabolites.
Modulating the microbiota through interventions such as probiotics, prebiotics, FMT, and dietary changes has proven effective in improving cancer outcomes, particularly in gastrointestinal cancers. These strategies help to protect the GI environment, reduce tumor burden, and enhance the efficacy of anticancer treatments. Furthermore, novel approaches, including culturomics, metagenomic sequencing, and genetic-based therapies, hold great promise for advancing microbiome research and improving cancer care.
The future of GI cancer treatment lies in the integration of cutting-edge microbiome research with clinical applications. Continued investment in research, technology, and interdisciplinary collaboration will be essential to fully harness the potential of the microbiota in cancer prevention and treatment.
Author Biography
Dr. Eshika Dubey, an MBBS doctor currently working in Paediatrics at HBT Cooper Municipal Hospital. She has experience in Nuclear Medicine from HN Reliance Hospital and aspires to specialize in Radiology or Dermatology in the UK. She is a graduate of MGM Medical College and is interested in unique cases, research and writing.