Source: www.Medscape.Com
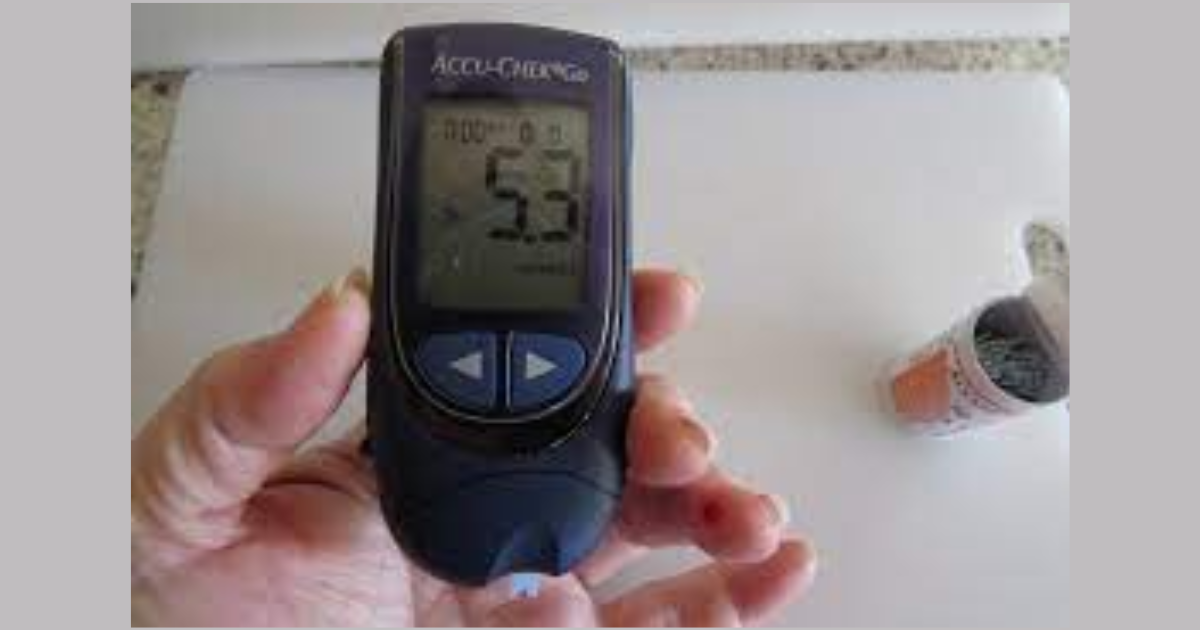
In a prospective, six month, single-arm study with use of a novel clinical-decision support tool and shared decision-making in 94 patients above 65 years old with type 2 diabetes managed in a primary care practice and at high risk for hypoglycemic episodes led to a 46% decrease in the number of at-risk patients and discontinuation of hypoglycemic agents in 20% patients.
The HypoPrevent study was done in a Pennsylvania primary care centre where 94 enrolled patients had type 2 diabetes and were at risk for hypoglycemia because of treatment with sulfonylureas or insulin with HbA1c of less than 7%. Patients averaged 74 years old, 57% were women, 955 were white, 61% had diabetes for more than 10 years, 51% were on insulin, 48%had chronic kidney disease and 47% were on sulfonylurea. Out of 94 patients, 80 completed all three study visits.

So, the patients and clinicians used a new hypoglycemia reduction clinical-decision support tool developed by the Endocrine Society and a healthcare consulting company. Resultantly, 20% patients reduced their dose of or discontinued insulin or sulfonylurea. In patients with both baseline and follow-up A1 measures, A1c rose from 6.29% at baseline to 6.82%. 50 patients set an A1c goal and had a timely follow-up A1c measurement and in this subgroup, the number of patients at risk for hypoglycemia decreased significantly by 46%. This proves that this decision support tool can be effectively used in primary care practice with positive results.
The drawback of this study clearly lies in the fact that there was lack of control group which makes it impossible to conclusively determine whether the intervention led to the observed increases in A1c levels nor can the study exclude regression to the mean as the cause for lowered A1c levels. The study received funding from Lilly, Merck, Abbott, Sanofi and Novo Nordisk. This study is published in the Journal of the American Geriatrics Society.